Our Services

LASIK
LASIK at Lusk Eye Specialists will not only change the way you SEE. It will change the way you LIVE! From your first consultation for laser eye surgery at Lusk Eye Specialists to the final post-operative follow-up, you’ll feel The Lusk Difference in action.

Cataract Surgery
For 40 years, the surgeons at Lusk Eye Specialists have set the standard for cataract care in the Ark-La-Tex. From pioneering the latest breakthroughs in laser and lens implant technology to providing more affordable cataract surgery options with our dedicated outpatient surgery center, Lusk Eye Specialists remain the clear choice for restoring your vision through cataract surgery.

Glaucoma Treatment
At Lusk Eye Specialists, we know that glaucoma can be a frightening diagnosis. You may not know how severe your glaucoma is, or what your options are for treatment. And, you may not be sure who to trust with your ongoing eye health and vision while managing this disease. The entire Lusk team is here to be your resource when you’re looking for effective management and treatment of your glaucoma, giving you the peace of mind that you deserve when you have a complex eye condition.

Dry Eye
Dry eye is rapidly becoming more common across the US, and Shreveport-Bossier is no exception. As we have increased our dependence on digital devices in our daily lives, dry eye symptoms are showing up for more and more patients. Sometimes these symptoms can come about so slowly that patients don’t even realize that their eyes are dry. They assume their discomfort is normal because of the hours they are spending on the computer or just a part of getting older.

Cornea Transplant

Eye Conditions
Lusk Eye Specialists is proud to provide compassionate solutions for a wide range of eye conditions. Our skilled team uses advanced technology to treat and manage cataracts, refractive vision and corneal issues, glaucoma, diabetic retinopathy, age-related macular degeneration (AMD), keratoconus, conjunctivitis, and more.

ICL
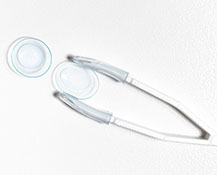
Scleral Lenses
These specialty lenses are the perfect option for patients who want the freedom that contact lenses provide but have a hard-to-fit prescription, severe dry eye symptoms, or corneal issues. Scleral lenses are a type of rigid gas permeable (RGP) lenses that are significantly larger than standard contact lenses and rest on the sclera, or the whites of your eyes, as opposed to your corneas.
LEARN MORE

Refractive Lens Exchange (RLE)